Part 1 - The Histopathology view
1. Indications for biopsy
Indications for gastrointestinal biopsy may be based on the clinical presentation of the animal, laboratory test results or other techniques such as imaging
Indications for GIT biopsy

2. GIT biopsy – Types, pros and cons
Table 1. Endoscopic biopsies
PROS
Fast and minimally invasive
AlLows direct evaluation of the GIT mucosa, particularly oesophagus, stomach, duodenum, ileum & colon
Allows potentially large numbers of samples to be taken at different levels of GIT
Samples are appropriate for histopathology as well as other techniques, such as immunohistochemistry, PARR and fluorescent in situ hybridisation (FISH)‡
CONS
Generally only captures mucosa and sometimes superficial submucosa, but not the wall
Lesions are often patchy in the GIT and this technique is better suited to more diffuse lesions
Cannot reach jejunum or distal duodenum in large dogs
Some samples are too hard to sample (e.g. carcinomas)
‡ Immunohistochemistry, PARR and FISH can be run on formalin fixed, paraffin embedded tissue
Table 2. Full-thickness biopsies
PROS
Any lesion can be sampled regardless of site, texture, size or consistency
More appropriate when evaluation of the intestinal submucosa and/or muscular wall is important, particularly for evaluation of neoplasia
May allow resection and cure of local disease
Allows assessment of the serosa and more of the GI tract, including less accessible areas (jejunum)
Samples are appropriate for histopathology as well as other techniques (see Table 1)
CONS
Much more invasive requiring abdominal surgery, hospitalisation and longer convalescence – leads to increased costs
Cannot see the mucosa
Slight risk of wound breakdown and peritonitis
Increased risk of post-operative morbidity
Example images of each type are present at the end
Special case: Enterectomy
Most of the time, the enterectomy samples we receive are localised lesions detected at surgery, on palpation or via imaging, the aim being to achieve complete removal and potentially cure. Examples include areas of ulceration/perforation, neoplasms, other space-occupying lesions, and intussusception sites.
Fig 2 shows the general approach to processing such specimens.
More samples may be required if considering copper analysis and culture, in addition to histopathology. In such circumstances, five samples are considered “adequate”5.
Collect as much tissue as possible; place different sites in different pots and label them.
Best to avoid lobe tips as they may be less representative or even misleading.
Fig 2. Intestinal resection with mass. Red boxes depict areas sampled, allowing evaluation of margins.

3. What to sample and how to send
Endoscopic samples: Sample stomach (fundus and pylorus), duodenum, ileum and colon depending on presentation. By taking eight good quality samples from each site, this will help to maximise diagnostic value, i.e. there will always be some samples that are not sufficiently deep, suboptimally oriented or artefactually crushed, but more numerous samples help to overcome this4.
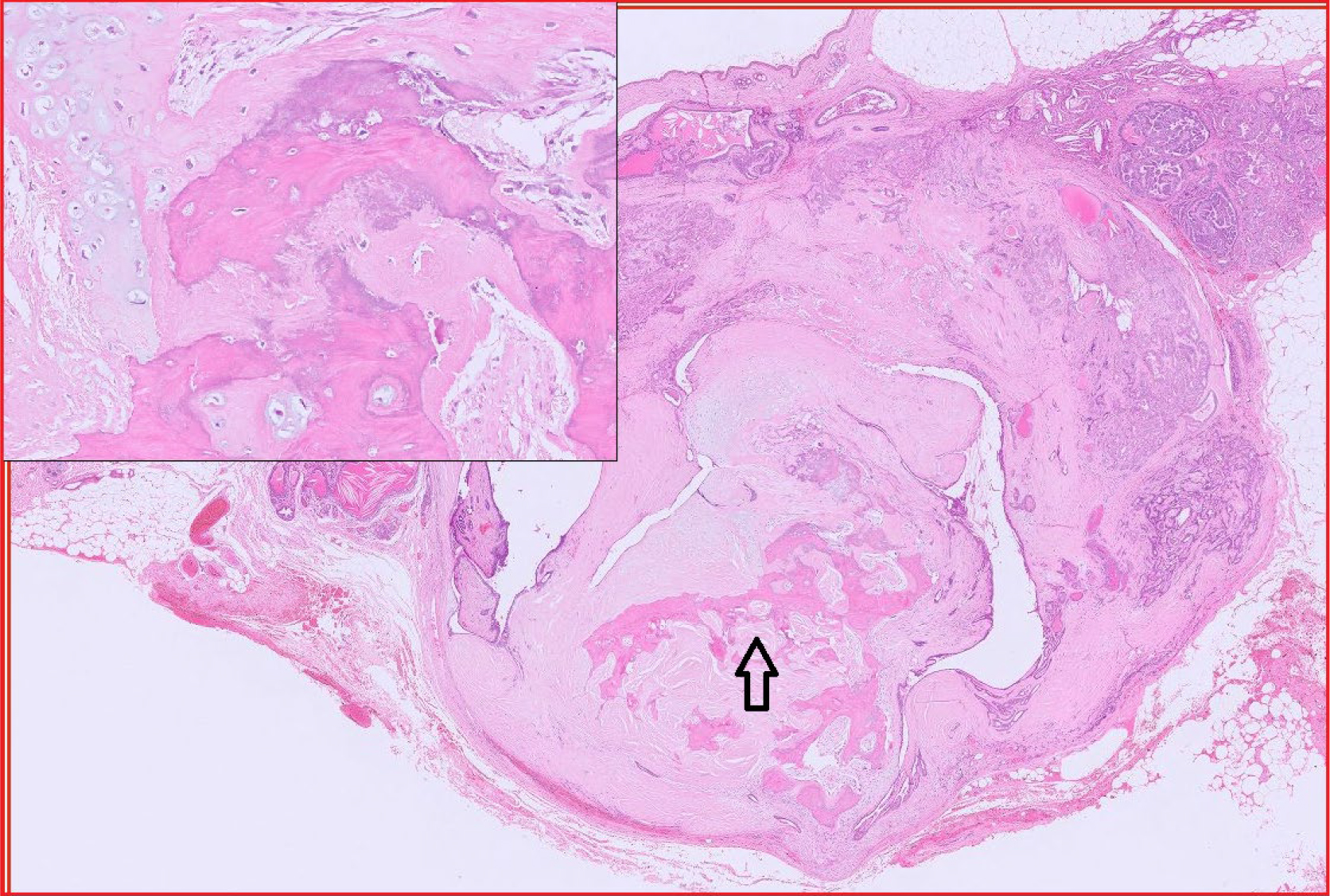
Full-thickness samples: These are the best samples to send if you are concerned about neoplasia or if there is suspicion of lesions in the submucosa or muscular wall. Many lesions are within the deep mucosa, crypts or submucosa so superficial samples may not be diagnostic. Adenocarcinoma of the GIT is often diagnosed based on invasive lesions in the submucosa or wall – endoscopic samples limited to the mucosa may miss the lesion (see Fig. 3, an example of invasive rectal adenocarcinoma).
How to send: Samples should be sent in 10% neutral buffered formalin. While samples can be shaken from the biopsy forceps directly into the formalin, studies have shown that better results are achieved by mounting the samples on a support medium, e.g. foam or cellulose acetate paper or filter paper5. They are also less likely to be overlooked. At our laboratory we can receive samples free floating in formalin, on foam, or on paper. Our preference is CellSafe6, a more secure tissue cassette with extra fine mesh. This sometimes comes with compartments so you can send multiple levels of GIT in the same cassette. Once samples are secured within the cassette, it is placed in a pot of formalin.
Please send different sites in different labelled pots. Stomach samples can go in the same pot unless you are concerned about a particular area in the stomach, e.g. you spot a lesion. Then you should send them separately labelled.
Fig 3. Rectal adenocarcinoma in a dog.

4. Tips
- Be as gentle as possible – GI tissue is very prone to crush artefact and this can result in a non-diagnostic sample. Duodenum is particularly prone to this4. Factors affecting quality include the experience of the operator; size and type of forceps used (can affect sample depth and size); and method of handling (e.g. mounted samples are of better quality compared to free floating). The laboratory obviously also has a responsibility to handle samples carefully.
- Samples of very good quality are usually very pale or white grossly. They should be long rather than round and should have a consistency typical of tissue such as skin. If they are gelatinous they are more likely to be non-diagnostic and only consist of mucus.
- Please bear in mind that severity of inflammatory lesions histologically do not correlate well with severity of clinical signs. Histopathology also cannot distinguish inflammatory bowel disease from other conditions such as food or antimicrobial responsive enteropathies, endoparasitism or bacterial overgrowth. A diagnosis of IBD is not solely histological; it requires consideration of other factors such as other clinical signs (vomiting, diarrhoea, weight loss), exclusion of systemic disease, response to dietary alterations or medications, and response to anti-inflammatories or immune modulators2.
- We sometimes receive mesenteric lymph nodes from animals with a history of alimentary disease, particularly cats. Lymph node alone very rarely leads to a diagnosis, unless there is lymphoma. There may be reactive hyperplasia but this is non-specific and only indicates antigenic stimulation. Since it takes some effort to sample lymph node in this way, it makes sense to send samples of GIT as well.
5. Relevant clinical information
The following information is extremely helpful when we are assessing gastrointestinal biopsies:
- Signalment
- Onset and progression of clinical signs
- Any gross lesions detected; their distribution if known (focal, diffuse, multifocal)
- Areas sampled
- Results of other tests
- Brief summary of treatment and any response



References
- Spillman T (2007) Endoscopy of the Gastrointestinal Tract: When is it Really Indicated. World Small Animal Veterinary Association World Congress Proceedings. Sydney, Australia.
- Rychlik A and Kaczmar E (2020) Endoscopic Biopsies and Histopathological Findings in Diagnosing Chronic Gastrointestinal Disorders in Dogs and Cats. Vet Med Int. https://doi.org/10.1155/2020/8827538.
- Mansell and Willard (2003) Biopsy of the gastrointestinal tract. Vet Clin Small Anim 33:1099-1116.
- Willard MD et al (2001) Quality of tissue specimens obtained endoscopically from the duodenum of dogs and cats. JAVMA 219:474-479.
- Ruiz et al (2016) Comparison of 3 Handling Techniques for Endoscopically Obtained Gastric and Duodenal Biopsy Specimens: A Prospective Study in Dogs and Cats. J Vet Int Med 30: 1014-1021.
- https://www.cellpath.com/cellsafe-biopsy-capsule-white-ebe-0201-02a.html
Related Posts
Reference Guide to Fine Needle Aspiration of Skin Lesions: A Practical and Diagnostic Cornerstone in Veterinary Cytology
Reference Guide to Fine Needle Aspiration of Skin Lesions: A Practical and Diagnostic Cornerstone in…
Oncology Service Now Available – FREE for 3 months
Download a PDF
2026 Bank Holiday Information
Download a PDF
Organ of the Month: Muscle
Organ of the month: Muscle I. Muscle biopsy In this blog, we turn to muscle…